Principal Investigator : Jérôme Sohier (CR CNRS)
People Involved in the Project : Kardelen Durmaz (doctorante), Lucas Lemarié (Doctorant), Danielle Campiol Arruda (MCU), Jean-Paul Salvi (T)
Funding: ANR MICROVOICE, ANR GELIHPARBAL, FUI SPINEFLEX, PREMATURATION CNRS, CIFRE
Collaborations: Vincent Gache (INMG, Lyon), E. Courtial (ICBMS, Lyon), L. Bailly (3SR, Grenoble), C. Vezy (UTT, Troyes), Pr. L. Moroni (Université de Maastricht, Pays-Bas), Pr. M. Yacoub (Imperial college, London, Angleterre), NORAKER (Lyon), SEGULA.
Our research focuses on the development of biomaterials to highlight and guide cellular behavior, aiming to provide innovative and functional therapeutic solutions.
Bioactive Hydrogels and Modulation of Cellular Behavior
The specific structure and organization of the extracellular matrix (ECM) are essential components of the properties of native, regenerated, or healed tissues, particularly for soft tissues. Mimicking the fibrillar component of the ECM, as well as its hydrated part, appears fundamental to provide tools for understanding cellular reactions.
In this context, we have developed innovative and versatile hydrogels from standardized polyethylene glycol (PEG) molecules and lysine dendrimers (DGL). These biomaterials are biocompatible, highly modifiable in terms of chemical composition, surface charge, and mechanical properties, and allow for intrinsic interaction with cells.
. 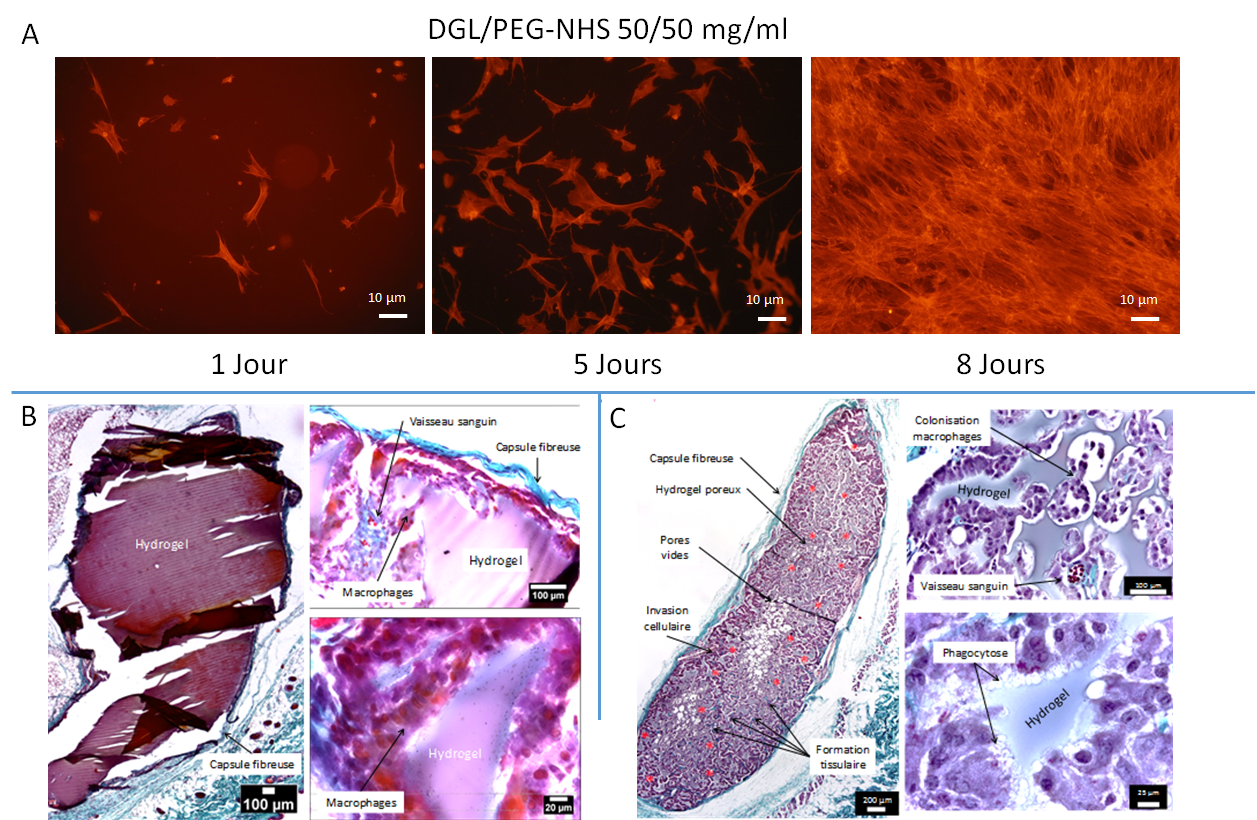
Morphology and Proliferation of Human Fibroblasts on the Surface of Hydrogels (A). Subcutaneous implantation of different hydrogels in mice: dense DGL/PEG-NHS hydrogels 50/50 mg/ml (D); DGL/PEG-NHS hydrogels 50/50 mg/ml containing 3.75 mg/ml of PDE and with 100-50 µm porosity (E). Masson’s trichrome staining indicating collagen in turquoise blue. Infiltrated blood vessels are indicated by a red asterisk.
The versatile properties of these hydrogels allow us to study the relationships between the behavior of different cell types and the mechanical and surface properties of the synthetic microenvironment.
Additionally, the direct aqueous formulation of these hydrogels allows for the incorporation of bioactive agents (molecules, proteins, or nanoparticles) and their controlled release.
They thus enable the definition of tissue engineering scaffolds and the formation of cellularized tissue models (in the case of muscle or skin) or acellular models (vocal cords or blood vessels).
Reconstruction of Skeletal Muscle Tissue in Large Defects
Deep muscle wounds lead to pathological fibrous healing, which no longer allows for functional contractility and often results in high disability.
Our project consisted of determining the optimal adhesion, migration, and proliferation properties on the hydrogel surface for murine or human myoblasts, in order to develop porous formulations supporting the fusion of myoblasts into myotubes and functional, contractile muscle fibers, whose sarcomeres are characterized by the presence of α-actinin striations. The three-dimensional microenvironment provided by these conditions also contributes to spontaneously inducing a pool of quiescent muscle stem cells, characterized by PAX7 expression and the absence of Ki67 and MyoD marker expression
. 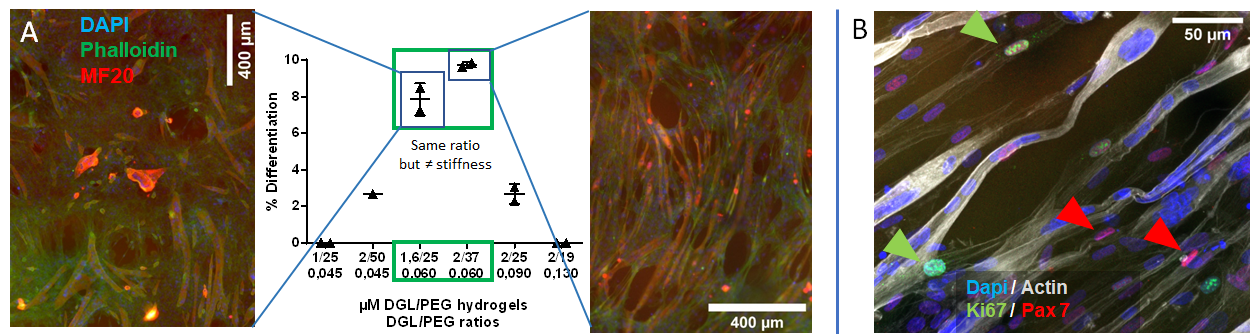
Influence of DGL/PEG Hydrogel Composition on the Fusion and Differentiation Capacity of Murine C2C12 After 6 Days of Culture, Visualized by Myosin Heavy Chain Staining (MF20) and the Number of Nuclei per Cell (A); and Population of Non-Proliferative Mononuclear Cells (Absence of Ki67 Expression) Expressing Pax7 Among the Myotubes Formed After 6 Days of Culture in Porous Hydrogels (B).
To evaluate the utility of our hydrogel in the context of muscle repair, we developed, for the first time, an effervescent and directly in-situ injectable formulation. This formulation allows for easy injection of the hydrogel directly into a muscle defect, while a highly interconnected porosity forms through effervescence concurrently with the injection. The porosity structure and its interconnection allow for significant and rapid colonization by myoblasts and muscle stem cells, as well as their fusion and differentiation into myotubes. Indicating a certain potential as a tissue formation scaffold, these porous hydrogels allow for vascularization, cellular invasion, and significant ECM deposition when injected in vivo subcutaneously. 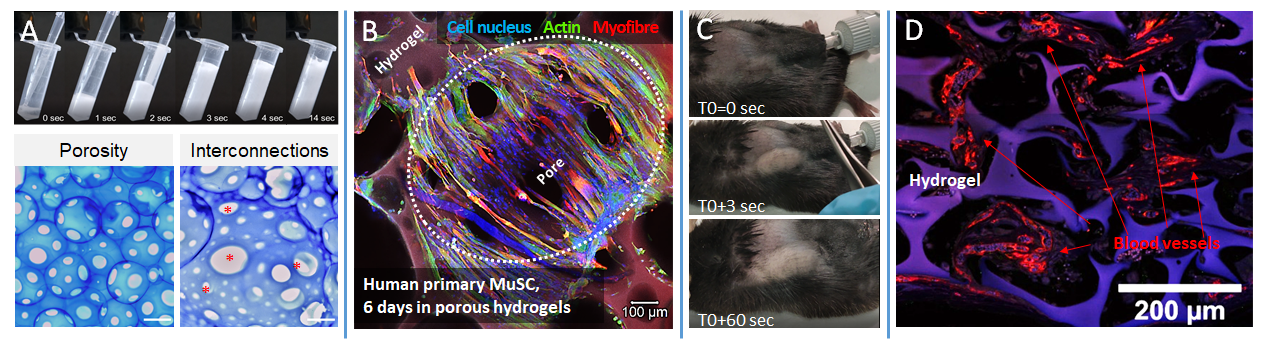
Effervescent Formulation of the Hydrogel (A) and Resulting Interconnected Porosity (A; scale 400 µm). Colonization of Resulting Porous Matrices by Human Myoblasts and Differentiation into Myofibers After 6 Days of Culture (B). Injection of Formulations Subcutaneously in a Murine Model (C) and Representative Example of Neovascularization Observed After 3 Weeks of Implantation (D). From Griveau, Lafont et al. 2021.
In vivo evaluation in large muscle defects in rats, currently underway, will allow us to validate our approach both histologically and functionally.
Modeling of Vocal Folds
LeThe vocal fold is a tissue with extraordinary properties, but little is known about it, particularly regarding the link between the microstructure of the vocal fold, its biomechanical behavior, and its remarkable vibratory properties.
By exploring the 3D fibrous architecture of the human vocal fold and designing mimetic fibrous/hydrogel composite materials with adjustable structural and biomechanical properties, we will be able to provide answers regarding the links between the vibromechanical performance of vocal tissues and their fibrous microstructures and surrounding matrices
. 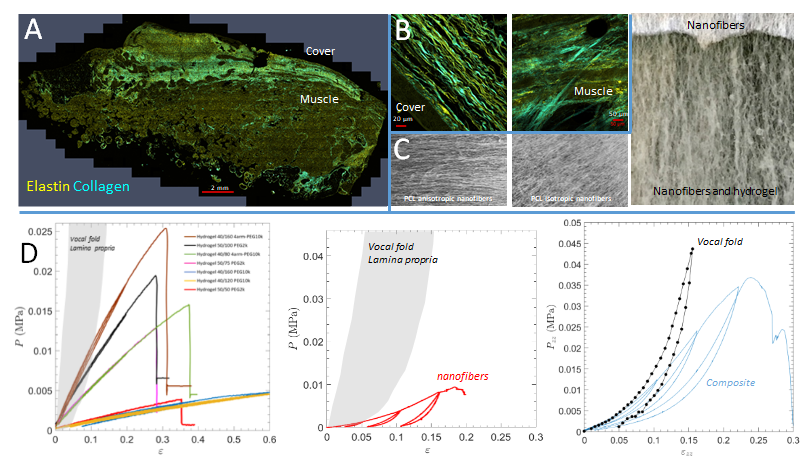
Two-photon confocal microscopy views of a human vocal fold showing the localization (A) and organization of the collagen and elastin network (B). Polycaprolactone nanofibers mimicking these architectures and allowing the formation of composites including the hydrogel (C). Mechanical properties under tension of hydrogel matrices, nanofibers, and composites, compared to those of human vocal folds (D).
Sélection of publications : A. Danoy, K. Durmaz, M. Paoletti, L. Vachez, A. Roul, J. Sohier, B. Verrier. Aqueous suspensions of Fuller’s earth potentiate the adsorption capacities of Paraoxon and improve skin decontamination properties. Journal of Hazardous Materials. 2022. 425: p. 127714. 10.1016/j.jhazmat.2021.127714 Ramirez Caballero S., Ferri Angulo, D., Debret, R., Granier, F., Marie, S., Lefèvre, F-X., Bouler, J-M, Despas, C., Sohier, J., Bujoli, B. Combination of biocompatible hydrogel precursors to apatitic calcium phosphate cements (CPCs): Influence of the in-situ hydrogel reticulation on the CPC properties. Journal of Biomedical Materials Research B: Applied Biomaterials. 2021 Jan;109(1):102-116. 10.1002/jbm.b.34685 Griveau, L., Lafont, M., le Goff, H., Drouglazet, C., Robbiani, B., Berthier, A., Sigaudo-Roussel, D., Latif, N., Le Visage, C., Gache, V., Debret, R., Weiss, P., Sohier, J. (2021). Design and characterization of an in vivo injectable hydrogel with effervescently generated porosity for regenerative medicine applications. Acta Biomaterialia: 2021 2022. 140: p. 324-337. 10.1016/j.actbio.2021.11.036 Carrancá, M., Griveau, L., Remoué, N., Lorion, C., Weiss, P., Orea, V., Sigaudo-Roussel, D., Faye, C., Ferri Angulo, D., Debret, R., Sohier, J. (2021) Versatile lysine dendrigrafts and polyethylene glycol hydrogels with inherent biological properties: in vitro cell behavior modulation and in vivo biocompatibility. Journal of Biomedical Materials Research A. 109(6):926-937; 10.1002/jbm.a.37083.



