Project leader : Jean-Daniel Malcor
Participant : D. Vertu-Ciolino, M. Pasdeloup, J-D Malcor, J. Lafont
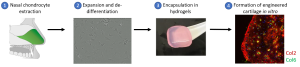
Nasal cartilage reconstruction represents a major clinical challenge. The demand for replacing nasal cartilage defects following oncological, traumatic, or congenital resection, as well as for cosmetic rhinoplasty, is constantly increasing. Current surgical options are limited by difficulties in obtaining sufficient cartilage tissue without causing morbidity at the donor site, the duration and complexity of the surgical procedure, and the risk of infection.

In a tissue engineering approach, we therefore fabricated a highly biocompatible prosthetic nasal septum using a 3D-printed, non-resorbable porous biomaterial coated with a biocompatible hydrogel containing nasal chondrocytes. This resulted in the formation of engineered cartilage tissues that can be implanted in vivo for nasal septum reconstruction.
Furthermore, nasal chondrocytes have a greater proliferation capacity, produce a denser extracellular matrix, and are more resistant to inflammatory signals than articular chondrocytes. They can also be harvested via a minimally invasive biopsy of the nasal septum, which does not cause morbidity at the donor site. Thus, we are also exploring the reconstruction of articular cartilage using biomaterials containing nasal chondrocytes.
Selected publications :
1-Vertu-Ciolino D, Brunard F, Courtial EJ, Pasdeloup M, Marquette CA, Perrier-Groult E, Mallein-Gerin F, Malcor JD. (2024) Challenges in Nasal Cartilage Tissue Engineering to Restore the Shape and Function of the Nose. Tissue Eng Part B Rev. 2024 Dec;30(6):581-595. doi: 10.1089/ten.TEB.2023.0326. Epub 2024 Apr 17.
2-Perrier-Groult E, Pérès E, Pasdeloup M, Gazzolo L, Duc Dodon M, Mallein-Gerin F. Evaluation of the biocompatibility and stability of allogeneic tissue-engineered cartilage in humanized mice. PLoS One. 2019 May 20;14(5):e0217183. doi: 10.1371/journal.pone.0217183. eCollection 2019.
3- Dufour A, Buffier M, Vertu-Ciolino D, Disant F, Mallein-Gerin F, Perrier-Groult E. Combination of bioactive factors and IEIK13 self-assembling peptide hydrogel promotes cartilage matrix production by human nasal chondrocytes. J Biomed Mater Res A. 2019 Apr;107(4):893-903. doi: 10.1002/jbm.a.36612. Epub 2019 Jan 31.
4- Mayer N, Lopa S, Talò G, Lovati AB, Pasdeloup M, Riboldi SA, Moretti M, Mallein-Gerin F. (2016) Interstitial Perfusion Culture with Specific Soluble Factors Inhibits Type I Collagen Production from Human Osteoarthritic Chondrocytes in Clinical-Grade Collagen Sponges. PLoS One. 1;11(9):e0161479. PMID: 27584727. doi: 10.1371/journal.pone.0161479. eCollection 2016.
5- Perrier-Groult E, Pasdeloup M, Malbouyres M, Galéra P, Mallein-Gerin F. (2013) Control of collagen production in mouse chondrocytes by using a combination of bone morphogenetic protein-2 and small interfering RNA targeting col1a1 for hydrogel-based tissue-engineered cartilage. Tissue Eng. Part C. 19 : 652-664.
6- Durbec M, Mayer N, Vertu-Ciolino D, Disant F, Mallein-Gerin F, Perrier-Groult E. (2014) [Reconstruction of nasal cartilage defects using a tissue engineering technique based on combination of high-density polyethylene and hydrogel]. Pathol. Biol. 62 : 137-145.



